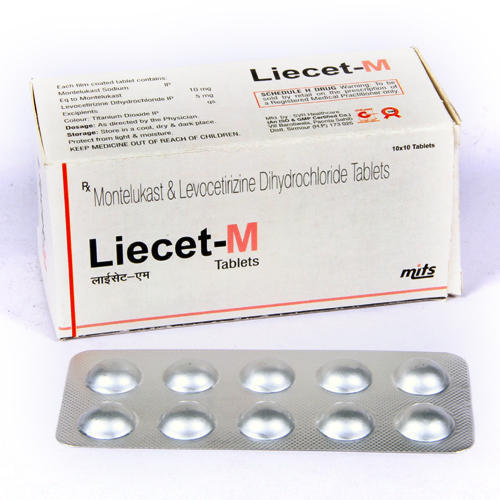
Montelukast and Levocetirizine Hydrochloride Tablets
Product Details:
- Drug Type Other Types
- Ingredients Montelukast 10 Levocetirizine Hydrochloride 10
- Physical Form Tablets
- Function Other
- Dosage As directed by doctor.
- Dosage Guidelines As directed by physician.
- Suitable For Women, Adults, Aged Person
- Click to View more
Montelukast and Levocetirizine Hydrochloride Tablets Price And Quantity
- 1 USD ($)/Box
- 100 Box
Montelukast and Levocetirizine Hydrochloride Tablets Product Specifications
- As directed by physician.
- Montelukast 10 Levocetirizine Hydrochloride 10
- Other Types
- Store at cool and dry place.
- Other
- Women, Adults, Aged Person
- As directed by doctor.
- Tablets
Montelukast and Levocetirizine Hydrochloride Tablets Trade Information
- Mumbai International Airport or By sea.
- Cash on Delivery (COD), Letter of Credit (L/C), Paypal, Western Union, Letter of Credit at Sight (Sight L/C), Cash Against Delivery (CAD), Cash Advance (CA), Telegraphic Transfer (T/T), Delivery Point (DP), Days after Acceptance (DA), Cash in Advance (CID), Cheque
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 10 Tablets in a strip, 10 strips in a box with product leaflet.
- Western Europe, Asia, Australia, North America, Eastern Europe, Central America, Middle East, South America, Africa
- Assam, Daman and Diu, Chandigarh, Nagaland, Uttarakhand, Dadra and Nagar Haveli, Lakshadweep, South India, Central India, North India, East India, West India, Andaman and Nicobar Islands, Andhra Pradesh, Arunachal Pradesh, Chhattisgarh, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Madhya Pradesh, Mizoram, Meghalaya, Manipur, Bihar, Punjab, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, West Bengal, Maharashtra, Delhi, Gujarat, Kerala, Uttar Pradesh, Odisha, Himachal Pradesh, All India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last many yearsSaintroyLifescienceis leading manufacture, export and supplierofMontelukast andLevocetirizine Hydrochloride Tablets from Surat, Gujarat, India. Montelukast and Levocetirizine Hydrochloride Tablets used inthe maintenance treatment of asthma. It is generally less preferred for thisuse than inhaled corticosteroids. Tags: Montelukast andLevocetirizine Hydrochloride Tablets manufacturer, Montelukast and LevocetirizineHydrochloride Tablets exporter, Montelukast and LevocetirizineHydrochloride Tablets supplier, and Montelukast and LevocetirizineHydrochloride Tablets ,whogmp approved Montelukast and Levocetirizine Hydrochloride Tablets manufacturerand supplier, Montelukast and Levocetirizine Hydrochloride Tablets distributor, Montelukast and LevocetirizineHydrochloride Tablets drop shipper, Montelukast and LevocetirizineHydrochloride Tablets 10/10mg Montelukastand Levocetirizine Hydrochloride Tablets 10/10mgsupplier, Montelukast andLevocetirizine Hydrochloride Tablets 10/10mg.
ISO/GMP/cGMP/WHOGMP/NAFDACapproved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approvedmanufacturingfacility in Gujarat, India.
Registration: Saintroy Lifescienceis interested inregistration of own brand or customer brands in ministryof health ofclients country and we also provide the products samples, COA, COPP, ACTD or CTDossier, DMF, Manufacturing license, Free sales certificate and other requireddocuments.
Export Country:Saintroy LifescienceMontelukast andLevocetirizine Hydrochloride Tablets exportto USA, Canada, Germany, Europe, France, Ghana, Nigeria, Kenya, Senegal,Sudan,UAE, Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile, Somalia, Philippines,Myanmar, Maldives, Thailand, Singapore, Malaysia, Hong Kong, China, Zimbabwe,Togo, Burkina Faso, Niger,Guinea Bissau, Congo, Democratic of republic, IvoryCosta, Oman, Guatemala, Nicaragua, Salvador, Qatar, Costa Rica, Panama, Cuba,Colombia, Bolivia, Argentine, Algeria, Guyana, Morocco, Jordan, Bahrain, Iran,Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.
Dual-Action Allergy Relief
Experience comprehensive symptom management, including runny nose, sneezing, and breathing difficulty, with the combined effect of Montelukast and Levocetirizine Hydrochloride. This formulation is ideal for adults and elderly patients seeking lasting relief from allergic reactions and mild asthma.
Safe Usage & Dosage
Always follow your doctor's prescription for dosage and use. The tablet must be taken exactly as recommended to ensure maximum benefit and minimize possible side effects. Consult your healthcare provider before starting, especially if you have other medical conditions.
Storage Guidelines for Potency
To preserve the quality and effectiveness of the tablets, store them in a cool, dry location, away from direct sunlight and moisture. Proper storage ensures that the medication remains stable and safe for use throughout its shelf life.
FAQ's of Montelukast and Levocetirizine Hydrochloride Tablets:
Q: How should Montelukast and Levocetirizine Hydrochloride Tablets be taken?
A: These tablets should be taken exactly as directed by your doctor or healthcare provider. Do not self-medicate or adjust your dose without medical advice.Q: What conditions do these tablets help treat?
A: Montelukast and Levocetirizine Hydrochloride Tablets are used to relieve symptoms of allergies, such as sneezing and runny nose, and to support the management of mild asthma in adults and elderly individuals.Q: When is the best time to take this medication?
A: The timing of your dose will be advised by your physician based on your condition and treatment plan. Typically, it can be taken once daily, but always follow your doctor's instructions.Q: Where should I store these tablets?
A: Store the tablets in a cool, dry place, away from heat and moisture. Keeping them in their original packaging will help protect their effectiveness.Q: What are the benefits of combining Montelukast and Levocetirizine?
A: This combination offers enhanced relief from allergy symptoms by targeting both inflammation and histamine response, leading to improved symptom control and better quality of life.Q: Who can use these tablets?
A: These tablets are suitable for women, adults, and older adults. Always consult your doctor before starting, to ensure suitability for your age and health condition.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+