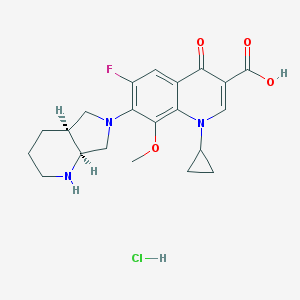
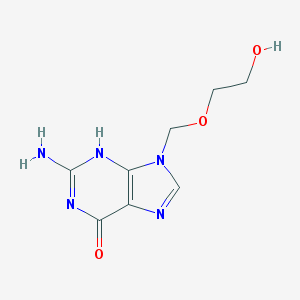
Moxifloxacin hcl
1-5 USD ($)
Product Details:
- Boiling point Not applicable / decomposes
- Particle Size Not more than 10 micron
- Heavy Metal (%) Not more than 0.001%
- Solubility Freely soluble in water, slightly soluble in ethanol
- Smell Odorless
- Taste Bitter
- Color White to pale yellow
- Click to View more
X
Moxifloxacin hcl Price And Quantity
- 1-5 USD ($)
- 100 Kilograms
Moxifloxacin hcl Product Specifications
- C21H24FN3O4HCl
- Approx. 225-228C
- Active Pharmaceutical Ingredient (API)
- Pharmaceutical Grade
- 2 years
- 4.0 6.0 (solution)
- Available on request
- 99%
- 186826-86-8
- 642-426-4
- Used for the treatment of bacterial infections
- Moxifloxacin HCl
- Not more than 0.5%
- 437.9 g/mol
- Store below 25C in a dry, well-closed container
- 29419090
- White to pale yellow
- Non-poisonous when used as directed
- Not more than 0.001%
- Not more than 10 micron
- Not applicable / decomposes
- White to pale yellow crystalline powder
- Freely soluble in water, slightly soluble in ethanol
- Odorless
- 1-Cyclopropyl-7-(2,8-diazabicyclo[4.3.0]non-8-yl)-6-fluoro-8-methoxy-4-oxo-quinoline-3-carboxylic acid hydrochloride
- Bitter
- Solid
Moxifloxacin hcl Trade Information
- Nhava sheva Port, Hajira Port, Mundra Port.A
- Cash Advance (CA), Letter of Credit (L/C), Western Union, Paypal, Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Cash in Advance (CID)
- 10000 Kilograms Per Month
- 6 Days
- Yes
- If order is confirmed we will reimburse the sample cost
- Australia, North America, South America, Eastern Europe, Middle East, Africa, Western Europe, Central America, Asia
- All India
- EUGMP/WHOGMP/GMP.
Product Description
Moxifloxacin Hydrochloride is the hydrochloride salt of a fluoroquinolone antibacterial antibiotic. Moxifloxacin binds to and inhibits the bacterial enzymes DNA gyrase (topoisomerase II) and topoisomerase IV, resulting in inhibition of DNA replication and repair and cell death in sensitive bacterial species.
Tags :- Moxifloxacin Manufacturer, Moxifloxacin Manufacturer inIndia, Moxifloxacin Supplier, Supplier in India, Moxifloxacin Distributor,Moxifloxacin Distributor in India, Moxifloxacin Merchant Exporter, MoxifloxacinExporter, Moxifloxacin Exporter in India, api, apis, PharmaceuticalIngredients, Active Pharmaceutical Ingredients, Moxifloxacin Bulk Drugsmanufacturer, Moxifloxacin Bulk Drugs, Moxifloxacin Bulk drugs in India,Moxifloxacin Api in India, Bulk Drugs, Bulk Drugs in India.
Superior Quality and Purity
This API maintains exceptional purity, with an assay range of 98.5%101.0% and total impurities capped at 0.5%. Rigorous identification via HPLC, IR, and UV ensures batch-to-batch consistency and compliance with global standards, establishing Moxifloxacin HCl as a trusted ingredient for pharmaceutical applications.
Comprehensive Safety Standards
Moxifloxacin HCl strictly adheres to pharmacopoeial criteria for chloride and heavy metals, ensuring less than 0.001% heavy metals and endotoxin levels below 0.5 EU/mg. Its non-poisonous nature, when properly used, adds an extra layer of safety for medical formulations, making it suitable for sensitive healthcare environments.
Secure Packaging and Storage
To guarantee stability and prevent contamination, Moxifloxacin HCl is packed in HDPE drums with double-laminated polyethylene bags. It should be stored below 25C in a dry, well-sealed container, securing the products integrity and extending its shelf life to a reliable two years.
FAQs of Moxifloxacin hcl:
Q: How is Moxifloxacin HCl typically used in pharmaceutical manufacturing?
A: Moxifloxacin HCl serves as a potent active pharmaceutical ingredient (API) in the production of antibacterial medications. It is incorporated into formulations aimed at treating a variety of bacterial infections, addressing pathogens through its broad-spectrum antimicrobial activity.Q: What are the purity and quality standards of Moxifloxacin HCl?
A: The product exhibits an assay between 98.5%101.0% by HPLC, boasts a purity of 99%, and maintains total impurities below 0.5%. Tests for identity (IR, UV, HPLC), chloride content, residual solvents, and heavy metals affirm compliance with global pharmacopoeial and ICH guidelines.Q: When should Moxifloxacin HCl be stored and handled with care?
A: It is essential to store Moxifloxacin HCl below 25C in a dry, well-closed container and away from direct sunlight or moisture. Proper handling during storage protects its chemical stability and ensures it remains effective throughout its two-year shelf life.Q: Where is Moxifloxacin HCl manufactured and how is it supplied?
A: Manufactured in India, Moxifloxacin HCl is supplied in HDPE drums lined with double-laminated polythene bags, each containing a net weight of 25 kg. This secure packaging minimizes contamination and guarantees product integrity during transit and storage.Q: What is the process for verifying the quality of Moxifloxacin HCl?
A: Quality assessment includes assays by HPLC, identity verification (IR/UV/HPLC), impurity profiling, and checks for chloride content, heavy metals, residual solvents, and endotoxin levels. These rigorous processes ensure each batch adheres to established pharmacopoeial limits.Q: What are the major benefits of using Moxifloxacin HCl as an API?
A: Moxifloxacin HCls high purity, broad-spectrum antibacterial action, and reliable safety profile make it a premium choice for pharmaceutical formulations. Its compliance with international standards further assures manufacturers of consistent, therapeutic efficacy.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Active Pharmaceutical Ingredients' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |