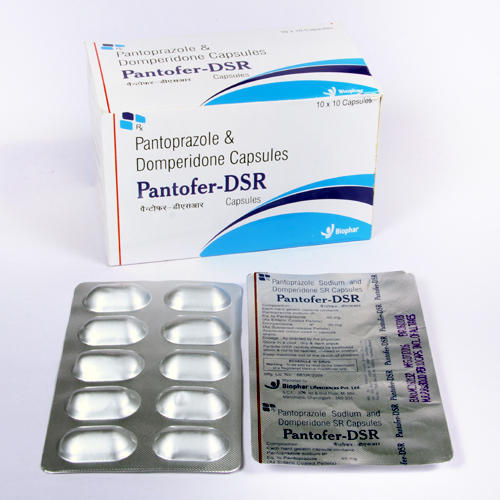
Pantoprazole Sodium and Domperidone Capsules
Product Details:
- Drug Type Other Types
- Ingredients Pantoprazole Sodium 40 Domperidone 30
- Physical Form Capsules
- Function Other
- Dosage As directed by doctor.
- Dosage Guidelines As directed by physician.
- Suitable For Women, Adults, Aged Person
- Click to View more
Pantoprazole Sodium and Domperidone Capsules Price And Quantity
- 100 Box
- 1 USD ($)/Box
Pantoprazole Sodium and Domperidone Capsules Product Specifications
- Pantoprazole Sodium 40 Domperidone 30
- As directed by doctor.
- Capsules
- Store at cool and dry place.
- Women, Adults, Aged Person
- As directed by physician.
- Other Types
- Other
Pantoprazole Sodium and Domperidone Capsules Trade Information
- Mumbai International Airport or By sea.
- Cash on Delivery (COD), Cash Advance (CA), Letter of Credit (L/C), Telegraphic Transfer (T/T), Western Union, Paypal, Letter of Credit at Sight (Sight L/C), Cash Against Delivery (CAD), Delivery Point (DP), Days after Acceptance (DA), Cash in Advance (CID), Cheque
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 10 Capsules in a strip, 10 strips in a box with product leaflet.
- Western Europe, Australia, Eastern Europe, Middle East, Central America, South America, Asia, North America, Africa
- Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Uttarakhand, Daman and Diu, Dadra and Nagar Haveli, Lakshadweep, Chhattisgarh, South India, North India, East India, Assam, Arunachal Pradesh, Bihar, Delhi, Gujarat, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Nagaland, Madhya Pradesh, Maharashtra, Mizoram, Meghalaya, Manipur, Punjab, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, Uttar Pradesh, West Bengal, All India, Andhra Pradesh, Kerala, Central India, Odisha, West India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last many yearsSaintroyLifescienceis leading manufacture, export and supplierofPantoprazole Sodiumand Domperidone Capsules from Surat, Gujarat, India. Pantoprazole Sodium and DomperidoneCapsules used for the treatment ofstomach ulcers, short-term treatment of erosive esophagitis due togastroesophageal reflux disease (GERD).
Tags: Pantoprazole Sodium and Domperidone Capsulesmanufacturer,Pantoprazole Sodium and Domperidone Capsules exporter,Pantoprazole Sodium and Domperidone Capsules supplier,and Pantoprazole Sodium and Domperidone Capsules , whogmp approved Pantoprazole Sodium andDomperidone Capsules manufacturer and supplier, Pantoprazole Sodiumand Domperidone Capsules distributor, Pantoprazole Sodium andDomperidone Capsules drop shipper, Pantoprazole Sodium andDomperidone Capsules 40/30mg Pantoprazole Sodium and DomperidoneCapsules 40/30mgsupplier, Pantoprazole Sodium andDomperidone Capsules 40/30mg.
ISO/GMP/cGMP/WHOGMP/NAFDACapproved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approvedmanufacturingfacility in Gujarat, India.
Registration: Saintroy Lifescienceis interested inregistration of own brand or customer brands in ministryof health ofclients country and we also provide the products samples, COA, COPP, ACTD or CTDossier, DMF, Manufacturing license, Free sales certificate and other requireddocuments.
Export Country:Saintroy LifesciencePantoprazole Sodiumand Domperidone Capsules export to USA, Canada, Germany, Europe,France, Ghana, Nigeria, Kenya, Senegal, Sudan,UAE, Kuwait, Dubai, Cameroon,Lebanon, Peru, Chile, Somalia, Philippines, Myanmar, Maldives, Thailand,Singapore, Malaysia, Hong Kong, China, Zimbabwe, Togo, Burkina Faso, Niger,GuineaBissau, Congo, Democratic of republic, Ivory Costa, Oman, Guatemala, Nicaragua,Salvador, Qatar, Costa Rica, Panama, Cuba, Colombia, Bolivia, Argentine,Algeria, Guyana, Morocco, Jordan, Bahrain, Iran, Egypt, Kyrgyzstan, Uzbekistan,Kazakhstan.
Dual-Action Relief for Gastrointestinal Health
These capsules harness the combined benefits of Pantoprazole Sodium, a proton pump inhibitor, and Domperidone, a prokinetic agent. Designed to relieve symptoms such as heartburn and indigestion, the formulation is especially suited for adults and the elderly. Its efficacy in improving digestive comfort makes it a practical solution for those suffering from frequent gastrointestinal issues.
Strict Quality and Storage Standards
Each batch of Pantoprazole Sodium and Domperidone Capsules is manufactured under rigorous quality controls. To preserve the integrity and therapeutic value of the medication, it's essential to store the capsules in a cool, dry place. This ensures the product maintains its potency and effectiveness during its shelf life.
FAQ's of Pantoprazole Sodium and Domperidone Capsules:
Q: How should Pantoprazole Sodium and Domperidone Capsules be taken for optimal results?
A: These capsules should be taken strictly as directed by your physician, typically before meals. The exact dosage and frequency depend on your individual health needs, so always follow your doctor's specific instructions.Q: What benefits do Pantoprazole Sodium and Domperidone offer when used together?
A: Pantoprazole Sodium reduces stomach acid production, while Domperidone helps stimulate movement in the digestive tract. Together, they help manage acid reflux, indigestion, and related symptoms more effectively than either ingredient alone.Q: When is the best time to take these capsules?
A: It is generally recommended to take these capsules before meals, but the precise timing and duration should follow your physician's guidance to ensure maximum benefit and absorption.Q: Where should I store these capsules?
A: Pantoprazole Sodium and Domperidone Capsules should be stored in a cool and dry place, away from direct sunlight and moisture, to preserve their effectiveness and safety.Q: What is the process for obtaining these capsules in India?
A: You can purchase these capsules from authorized dealers, suppliers, exporters, manufacturers, or wholesalers across India. It's important to obtain them with a prescription from a licensed healthcare provider.Q: Who are these capsules suitable for?
A: These capsules are formulated for women, adults, and aged individuals who require gastrointestinal support. However, they must be used under medical supervision to ensure safety and appropriate treatment.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+