Active Pharmaceutical Ingredients
1 USD ($)
Product Details:
- HS Code 2942
- EINECS No Varies with the API
- Melting Point Varies with API
- Smell Odorless or characteristic
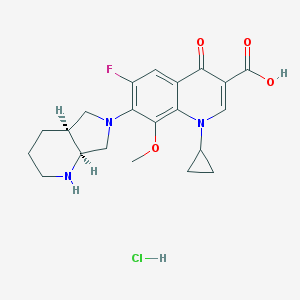
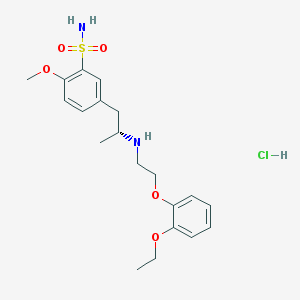
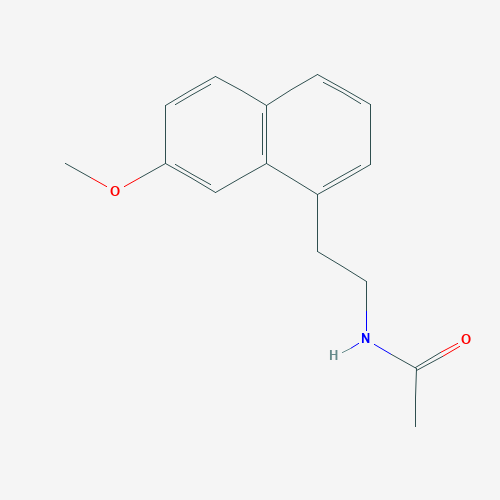
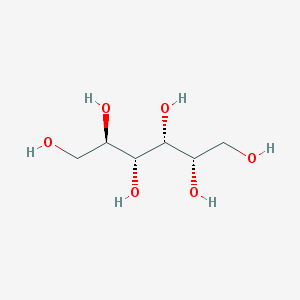
- Structural Formula Varies with API
- Boiling point Varies with API
- Molecular Weight Varies with API
- Click to View more
X
Active Pharmaceutical Ingredients Price And Quantity
- 1 USD ($)
- 50 Kilograms
Active Pharmaceutical Ingredients Product Specifications
- Cool, dry, well-ventilated area
- Non-poisonous under normal conditions
- Active Pharmaceutical Ingredients
- Varies with API
- Pharmaceutical Grade
- <0.001%
- Varies with API
- 90% below 10 microns
- Pharmaceutical Formulation, Drug Manufacturing
- 99% min
- Tasteless or slightly bitter
- Varies as per specific ingredient
- Varies depending on individual API
- Max 1.0%
- Powder
- White to off-white
- 5.0 - 7.5
- 2-5 years depending on API
- Varies with API
- Varies with API
- Pharmaceutical Raw Material
- Varies with API
- Varies with the API
- White or off-white powder
- 2942
- Odorless or characteristic
- Varies with API
- Synthetic or Fermentation (as per API)
- < 0.2%
- 0.5% (by KF)
- HDPE drum with double liner or as specified
- As per standard for API
- Complies with pharmacopeial specifications
- Stable under recommended storage conditions
- Complies with pharmacopeia
- IR, HPLC or as per pharmacopeial monograph
- Complies with ICH guidelines
Active Pharmaceutical Ingredients Trade Information
- Nhavasheva Port, Mundra Port, Hajira Port.
- 10000 Kilograms Per Month
- 7 Days
- If order is confirmed we will reimburse the sample cost
- 25 kg drum packing.
- South America, Eastern Europe, Middle East, Western Europe, Asia, Central America, North America, Australia, Africa
- WHOGMP/GMP/EUGMP/USFDA
Product Description
Active Pharmaceutical Ingredients
We Saintroy Life science established in 2016 in hub of Pharmaceutical products ,Gujarat ,India.We offers all types of Active Pharmaceutical Ingredients from Surat.We also support all types of documents like COA, DMF with WHOGMP/EUGMP certified manufacturer at affordable rates .Providing quality and on time delivery is our company's basic moto.
Stringent Quality Assurance
Each Active Pharmaceutical Ingredient is rigorously tested to comply with ICH guidelines for residual solvents and pharmacopeial limits for microbial content and endotoxins. High-performance techniques such as IR and HPLC ensure reliable identification and quality. Standard testing also covers specific rotation, water content, heavy metals, and related substances, guaranteeing safe and consistent use in medicinal applications.
Versatile Manufacturing Methods
APIs can be synthesized using traditional chemical reactions or produced via fermentation processes, depending on the nature of the ingredient. These controlled methods ensure consistent purity and structural integrity across every batch. With flexible production, we can cater to a range of pharmaceutical requirements, from small molecules to complex biologics.
Superior Packaging and Stability
All APIs are packaged in HDPE drums with double liners or as specified per client requirements. This high-standard packaging preserves the chemical stability and physical quality of the powder, preventing contamination and degradation during transit and storage. When stored in cool, dry, well-ventilated environments, the shelf life ranges from two to five years, depending on the specific API.
FAQs of Active Pharmaceutical Ingredients:
Q: How are Active Pharmaceutical Ingredients tested for quality and compliance?
A: APIs are tested using state-of-the-art techniques such as Infrared Spectroscopy (IR), High-Performance Liquid Chromatography (HPLC), and methods specified in pharmacopeial monographs. These tests ensure the product meets standards for purity, potency, residual solvents, microbial limits, endotoxins, and other quality parameters.Q: What are the recommended storage conditions for Active Pharmaceutical Ingredients?
A: APIs should be stored in a cool, dry, and well-ventilated area, ideally in tightly sealed containers such as HDPE drums with double liners. Proper storage ensures stability and maintains the shelf life, usually ranging from two to five years.Q: When is the ideal time to use these APIs in pharmaceutical manufacturing?
A: These APIs are best used during the formulation and production phase of medicinal products. Timing depends on your manufacturing processes and batch requirements, ensuring the highest efficacy and integrity of finished pharmaceuticals.Q: Where do you source and supply your Active Pharmaceutical Ingredients?
A: APIs are manufactured and supplied in India, catering to national and international markets as a dealer, distributor, exporter, manufacturer, supplier, trader, and wholesaler. Sourcing follows strict regulatory and quality protocols.Q: What are the main benefits of choosing these pharmaceutical-grade APIs?
A: Key benefits include high purity (minimum 99%), compliance with international quality standards, extended shelf life, excellent microbial safety, and secure packaging. These APIs support consistent and safe drug production.Q: How are the APIs packaged to prevent contamination or degradation?
A: APIs are securely packed in HDPE drums with double liners, minimizing exposure to environmental factors like moisture and contaminants. This packaging method preserves the physical and chemical stability of the product until final use.Q: What is the manufacturing process for APIs and how is it chosen?
A: APIs are manufactured synthetically or by fermentation, determined by the compounds chemical nature and regulatory requirements. The selected method ensures optimal yield, purity, and compliance with pharmacopeial and ICH guidelines.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email