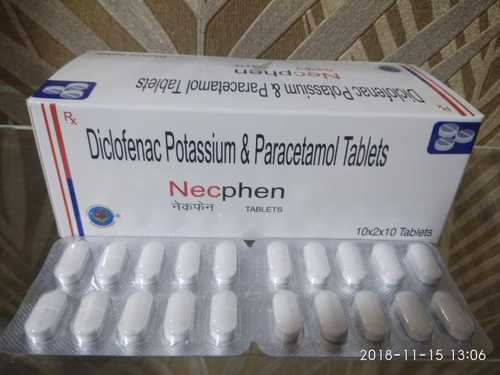
Diclofenac Potasium & Paracetamol Tablets
Product Details:
- Drug Type Other Types
- Ingredients Diclofenac Potasium 50 Paracetamol 325
- Physical Form Tablets
- Function Other
- Dosage As directed by doctor.
- Dosage Guidelines As directed by physician.
- Suitable For Teenagers, Women, Aged Person
- Click to View more
Diclofenac Potasium & Paracetamol Tablets Price And Quantity
- 1 USD ($)/Box
- 100 Box
Diclofenac Potasium & Paracetamol Tablets Product Specifications
- Tablets
- Teenagers, Women, Aged Person
- As directed by doctor.
- Diclofenac Potasium 50 Paracetamol 325
- Other
- Other Types
- As directed by physician.
- Store at cool and dry place.
Diclofenac Potasium & Paracetamol Tablets Trade Information
- Mumbai International Airport or By sea.
- Cash on Delivery (COD), Cash Advance (CA), Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Paypal, Letter of Credit (L/C), Western Union, Cash Against Delivery (CAD), Delivery Point (DP), Days after Acceptance (DA), Cash in Advance (CID), Cheque
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 10*2 Tablets in a strip, 10 strips in a box with product leaflet.
- Western Europe, Asia, Australia, North America, Eastern Europe, Middle East, Africa, Central America, South America
- Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Uttarakhand, Daman and Diu, Dadra and Nagar Haveli, Lakshadweep, South India, Central India, North India, East India, West India, Andhra Pradesh, Assam, Arunachal Pradesh, Bihar, Delhi, Gujarat, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Kerala, Madhya Pradesh, Maharashtra, Mizoram, Meghalaya, Manipur, Punjab, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, Uttar Pradesh, West Bengal, Nagaland, Odisha, Chhattisgarh, All India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Diclofenac Potasium & Paracetamol Tablets used nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammatory diseases such as gout. It is taken by mouth, rectally in a suppository, used by injection, or applied to the skin.
Tags: Diclofenac Potasium & Paracetamol Tablets manufacturer, Diclofenac Potasium & Paracetamol Tablets exporter, Diclofenac Potasium & Paracetamol Tablets supplier, Diclofenac Potasium & Paracetamol Tablets, Diclofenac Potasium & Paracetamol Tablets , who gmp approved Diclofenac Potasium & Paracetamol Tablets manufacturer and supplier, Diclofenac Potasium & Paracetamol Tablets distributor, Diclofenac Potasium & Paracetamol Tablets drop shipper, Diclofenac Potasium & Paracetamol Tablets 50 mg/325 mg, Diclofenac Potasium & Paracetamol Tablets 50 mg/325 mg supplier, Diclofenac Potasium & Paracetamol Tablets 50 mg/325 mg.
ISO/GMP/c-GMP/WHOGMP/NAFDAC approved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approved manufacturing facility in Gujarat, India.
Registration: Saintroy Lifescience is interested in registration of own brand or customer brands in ministry of health of clients country and we also provide the products samples, COA, COPP, ACTD or CTD Dossier, DMF, Manufacturing license, Free sales certificate and other required documents.
Export Country : Saintroy Lifescience Diclofenac Potasium & Paracetamol Tablets export to USA, Canada, Germany, Europe, France, Ghana, Nigeria, Kenya, Senegal, Sudan, UAE ,Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile, Somalia, Philippines, Myanmar, Maldives, Thailand, Singapore, Malaysia, Hong Kong, China, Zimbabwe, Togo ,Burkina Faso, Niger, Guinea Bissau, Congo, Democratic of republic, Ivory Costa ,Oman, Guatemala, Nicaragua, Salvador, Qatar, Costa Rica, Panama, Cuba, Colombia, Bolivia, Argentine, Algeria, Guyana, Morocco, Jordan, Bahrain, Iran, Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.
Dual-Action Pain Relief
By combining Diclofenac Potassium and Paracetamol, these tablets provide efficient pain relief and reduce inflammation. The synergy between the two ingredients helps manage acute discomfort and swelling, making it a reliable option for both chronic and sudden pain episodes.
Suitable for Different Age Groups
This medication is carefully formulated to be safe for aged people, teenagers, and women when used as per medical guidance. It is versatile in application, addressing a broad spectrum of pain-related conditions without compromising safety for the indicated groups.
Available Across India
Diclofenac Potassium & Paracetamol Tablets are widely available through multiple channels, including dealers, distributors, exporters, manufacturers, suppliers, traders, and wholesalers throughout India. This ensures easy access for both healthcare providers and patients.
FAQ's of Diclofenac Potasium & Paracetamol Tablets:
Q: How should Diclofenac Potassium & Paracetamol Tablets be taken for best results?
A: These tablets should be taken orally with water, exactly as your physician prescribes. Do not self-medicate or exceed the recommended dosage.Q: What conditions can be managed using this medication?
A: It is commonly used to alleviate pain and inflammation in conditions such as arthritis, muscle aches, headaches, and other painful disorders.Q: When is the right time to take these tablets?
A: Take the tablets at the intervals advised by your doctor, which may depend on your specific condition and response to treatment. Always follow your prescribed schedule.Q: Where should I store Diclofenac Potassium & Paracetamol Tablets?
A: Store these tablets in a cool, dry place away from direct sunlight and moisture to maintain their efficacy.Q: What is the process to obtain these tablets in India?
A: You can acquire them through various authorized dealers, distributors, suppliers, or pharmacists, making the tablets accessible across the country.Q: What are the benefits of combining Diclofenac Potassium and Paracetamol?
A: The combination offers enhanced pain relief and anti-inflammatory effects, providing a faster and broader spectrum of symptom management compared to single-agent therapies.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+